Source of Irons
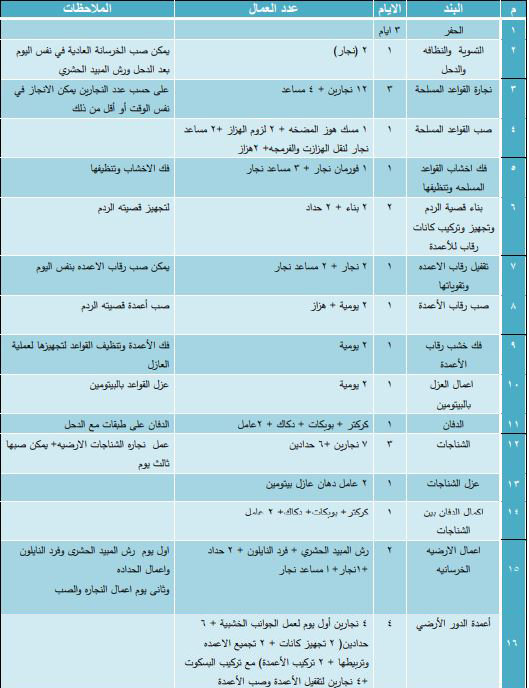
المرافق وأعمال الموقع
Iron
Background
Iron is one of the most common elements on earth. Nearly every construction of man
contains at least a little iron. It is also one of the oldest metals and was first fashioned
into useful and ornamental objects at least 3,500 years ago.
Pure iron is a soft, grayish-white metal. Although iron is a common element, pure iron is
almost never found in nature. The only pure iron known to exist naturally comes from
fallen meteorites. Most iron is found in minerals formed by the combination of iron with
other elements. Iron oxides are the most common. Those minerals near the surface of
the earth that have the highest iron content are known as iron ores and are mined
commercially.
Iron ore is converted into various types of iron through several processes. The most
common process is the use of a blast furnace to produce pig iron which is about 92-
94% iron and 3-5% carbon with smaller amounts of other elements. Pig iron has only
limited uses, and most of this iron goes on to a steel mill where it is converted into
various steel alloys by further reducing the carbon content and adding other elements
such as manganese and nickel to give the steel specific properties.
History
Historians believe that the Egyptians were the first people to work with small amounts of
iron, some five or six thousand years ago. The metal they used was apparently
extracted from meteorites. Evidence of what is believed to be the first example of iron
mining and smelting points to the ancient Hittite culture in what is now Turkey. Because
iron was a far superior material for the manufacture of weapons and tools than any
other known metal, its production was a closely guarded secret. However, the basic
technique was simple, and the use of iron gradually spread. As useful as it was
compared to other materials, iron had disadvantages. The quality of the tools made
from it was highly variable, depending on the region from which the iron ore was taken
and the method used to extract the iron. The chemical nature of the changes taking
place during the extraction were not understood; in particular, the importance of carbon
to the metal’s hardness. Practices varied widely in different parts of the world. There is
evidence, for example, that the Chinese were able to melt and cast iron implements
very early, and that the Japanese produced amazing results with steel in small
amounts, as evidenced by heirloom swords dating back centuries. Similar
breakthroughs were made in the Middle East and India, but the processes never
emerged into the rest of the world. For centuries the Europeans lacked methods for
heating iron to the melting point at all. To produce iron, they slowly burned iron ore with
wood in a clay-lined oven. The iron separated from the surrounding rock but never quite
melted. Instead, it formed a crusty slag which was removed by hammering. This
repeated heating and hammering process mixed oxygen with the iron oxide to produce
iron, and removed the carbon from the metal. The result was nearly pure iron, easily
shaped with hammers and tongs but too soft to take and keep a good edge. Because
the metal was shaped, or wrought, by hammering, it came to be called wrought iron.
Tools and weapons brought back to Europe from the East were made of an iron that
had been melted and cast into shape. Retaining more carbon, cast iron is harder than
wrought iron and will hold a cutting edge. However, it is also more brittle than wrought
iron. The European iron workers knew the Easterners had better iron, but not the
processes involved in fashioning stronger iron products. Entire nations launched efforts
to discover the process.
The first known European breakthrough in the production of cast iron, which led quickly
to the first practical steel, did not come until 1740. In that year, Benjamin Huntsman took
out a patent for the melting of material for the production of steel springs to be used in
clockmaking. Over the next 20 years or so, the procedure became more widely
adopted. Huntsman used a blast furnace to melt wrought iron in a clay crucible. He then
added carefully measured amounts of pure charcoal to the melted metal. The resulting
alloy was both strong and flexible when cast into springs. Since Huntsman was
originally interested only in making better clocks, his crucible steel led directly to the
development of nautical chronometers, which, in turn, made global navigation possible
by allowing mariners to precisely determine their east/west position. The fact that he
had also invented modern metallurgy was a side-effect which he apparently failed to
notice.
Raw Materials
The raw materials used to produce pig iron in a blast furnace are iron ore, coke, sinter,
and limestone. Iron ores are mainly iron oxides and include magnetite, hematite,
limonite, and many other rocks. The iron content of these ores ranges from 70% down
to 20% or less. Coke is a substance made by heating coal until it becomes almost pure
carbon. Sinter is made of lesser grade, finely divided iron ore which, is roasted with
coke and lime to remove a large amount of the impurities in the ore. Limestone occurs
naturally and is a source of calcium carbonate.
Other metals are sometimes mixed with iron in the production of various forms of steel,
such as chromium, nickel, manganese, molybdenum, and tungsten.
The Ore Extraction and Refining Process
Before iron ore can be used in a blast furnace, it must be extracted from the ground and
partially refined to remove most of the impurities
Historically, iron was produced by the hot-blast method, or later, the anthracite furnace.
Either way, the fundamental activity in iron making involved a worker stirring small
batches of pig iron and cinder until the iron separated from the slag. Called “puddling,”
this was highly skilled work, but was also hot, strenuous, and dangerous. It required a
lot of experience as well as a hearty constitution. Puddlers were proud, independent,
and highly paid.
Puddlers founded the first trade union in the iron and steel industry, the Sons of Vulcan,
in Pittsburgh in 1858. In 1876, this union merged with three other labor organizations to
form the Amalgamated Association of Iron and Steel Workers. This was the union that
Andrew Carnegie defeated in the Homestead Strike of 1892, leaving the union in
shambles and the industry essentially unorganized until the 1930s
William S. Pretzer
Extraction
. 1 Much of the world’s iron ore is extracted through open pit mining in which the surface of the ground is removed by heavy machines, often over a very large
area, to expose the ore beneath. In cases where it is not economical to remove
the surface, shafts are dug into the earth, with side tunnels to follow the layer of
ore.
Refining
. 2 The mined ore is crushed and sorted. The best grades of ore contain over 60%
iron. Lesser grades are treated, or refined, to remove various contaminants
before the ore is shipped to the blast furnace. Collectively, these refining
methods are called beneficiation and include further crushing, washing with water
to float sand and clay away, magnetic separation, pelletizing, and sintering. As
more of the world’s known supply of high iron content ore is depleted, these
refining techniques have become increasingly important.
. 3 The refined ore is then loaded on trains or ships and transported to the blast
furnace site.
The Manufacturing Process
Charging the blast furnace
• 1 After processing, the ore is blended with other ore and goes to the blast
furnace. A blast furnace is a tower-shaped structure, made of steel, and lined
with refractory, or heat-resistant bricks. The mixture of raw material, or charge,
enters at the top of the blast furnace. At the bottom of the furnace, very hot air is
blown, or blasted, in through nozzles called tuye’res. The coke burns in the
presence of the hot air. The oxygen in the air reacts with the carbon in the coke
to form carbon monoxide. The carbon monoxide then reacts with the iron ore to form carbon dioxide and pure iron
Separating the iron from the slag
. 2 The melted iron sinks to the bottom of the furnace. The limestone combines
with the rock and other impurities in the ore to form a slag which is lighter than
the iron and floats on top. As the volume of the charge is reduced, more is
continually added at the top of the furnace. The iron and slag are drawn off
separately from the bottom of the furnace. The melted iron might go to a further
alloying process, or might be cast into ingots called pigs. The slag is carried
away for disposal.
Treating the gases
. 3 The hot gases produced in the chemical reactions are drawn off at the top and
routed to a gas cleaning plant where they are cleaned, or scrubbed, and sent back into the furnace; the remaining carbon monoxide, in particular, is useful to
the chemical reactions going on within the furnace.
A blast furnace normally runs day and night for several years. Eventually the
brick lining begins to crumble, and the furnace is then shut down for
maintenance.
Quality Control
The blast furnace operation is highly instrumented and is monitored continuously. Times
and temperatures are checked and recorded. The chemical content of the iron ores
received from the various mines are checked, and the ore is blended with other iron ore
to achieve the desired charge. Samples are taken from each pour and checked for
chemical content and mechanical properties such as strength and hardness.
Byproducts/Waste
There are a great many possible environmental effects from the iron industry. The first
and most obvious is the process of open pit mining. Huge tracts of land are stripped to
bare rock. Today, depleted mining sites are commonly used as landfills, then covered
over and landscaped. Some of these landfills themselves become environmental
problems, since in the recent past, some were used for the disposal of highly toxic
substances which leached into soil and water.
The process of extracting iron from ore produces great quantities of poisonous and
corrosive gases. In practice, these gases are scrubbed and recycled. Inevitably,
however, some small amounts of toxic gases escape to the atmosphere.
A byproduct of iron purification is slag, which is produced in huge amounts. This
material is largely inert, but must still be disposed of in landfills.
Ironmaking uses up huge amounts of coal. The coal is not used directly, but is first
reduced to coke which consists of almost pure carbon. The many chemical byproducts
of coking are almost all toxic, but they are also commercially useful. These products
include ammonia, which is used in a vast number of products; phenol, which is used to
make plastics, cutting oils, and antiseptics; cresols, which go into herbicides, pesticides,
pharmaceuticals, and photographic chemicals; and toluene, which is an ingredient in
many complex chemical products such as solvents and explosives.
Scrap iron and steel—in the form of old cars, appliances and even entire steel-girdered
buildings—are also an environmental concern. Most of this material is recycled,
however, since steel scrap is an essential resource in steelmaking. Scrap which isn’t
recycled eventually turns into iron oxide, or rust, and returns to the ground.
The Future
On the surface, the future of iron production—especially in the United States—appears
troubled. Reserves of high-quality ore have become considerably depleted in areas
where it can be economically extracted. Many long-time steel mills have closed.
However, these appearances are deceiving. New ore-enrichment techniques have
made the use of lower-grade ore much more attractive, and there is a vast supply of
that ore. Many steel plants have closed in recent decades, but this is largely because
fewer are needed. The efficiency of blast furnaces alone has improved remarkably. At
the beginning of this century, the largest blast furnace in the United States produced
644 tons of pig iron a day. It is believed that soon the possible production of a single
furnace will reach 4,000 tons per day. Since many of these more modern plants have
been built overseas, it has actually become more economical in some cases to ship
steel across the ocean than to produce it in older U.S. plants.